Enhancing Access to High‑Quality Pediatric Clinical Trials at CHEO
The Clinical Trial Access Core strengthens CHEO’s ability to deliver excellent, participant‑centred clinical research by providing centralized expertise, clinical research space, and support services. Our goal is to make it easier for children, youth, families, clinicians, researchers, and industry partners to access, partner, and participate in safe, high‑quality clinical trials.
By coordinating key clinical trial resources—including dedicated pediatric clinical trial space and equipment, specialized research nursing, and streamlined study start up and research procedures—the Clinical Trial Access Core helps accelerate discoveries and improve health outcomes for children and youth.
Clinical Investigation Unit
A Dedicated Pediatric Clinical Trial Environment
The Clinical Investigation Unit (CIU) is a dedicated clinical space within the Research Institute designed specifically for pediatric clinical trials. Purpose‑built with children and families in mind, the CIU incorporates features that promote comfort, engagement, and reduced stress for young research participants.
The CIU enables safe, high‑quality research visits through the expertise of its specialized staff, including experienced Clinical Trial Nurse Coordinators. Together, the team follows standardized procedures supported by institutional oversight, quality assurance processes, and study navigation to ensure consistency and safety across all visits.
The unit supports studies across all therapeutic areas and offers a child‑ and family‑centred environment that fosters positive research experiences and high‑quality data collection. Families can find the CIU using the CHEO digital map.
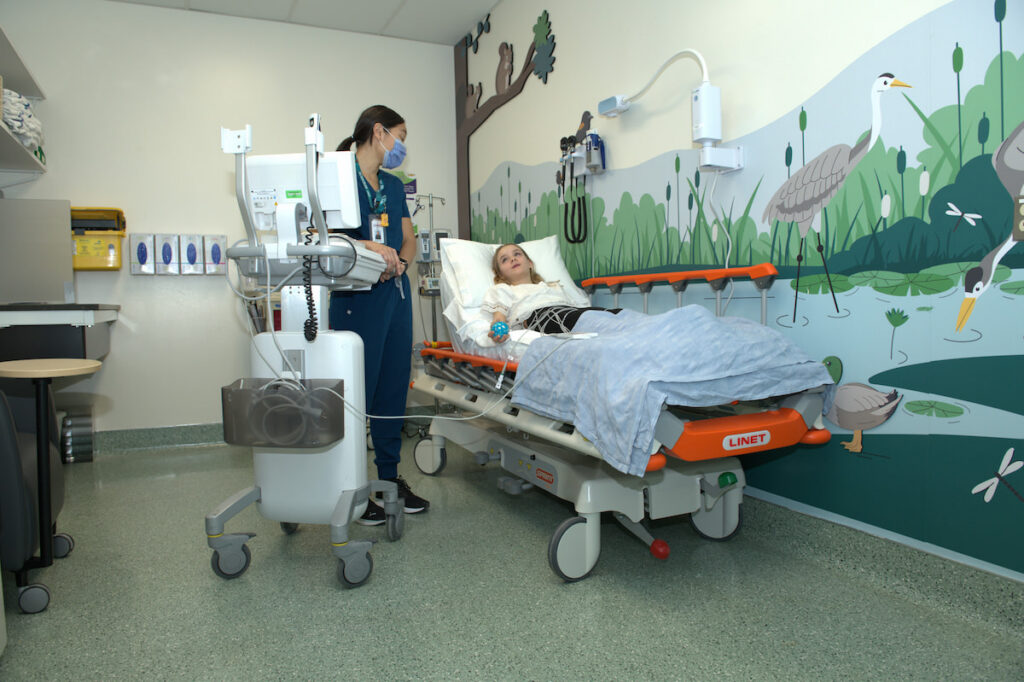
What the CIU Provides
Pediatric Clinical Trial Nursing
Our Clinical Trial Nurse Coordinators are experienced pediatric Registered Nurses with specialized expertise in conducting clinical trial procedures for infants, children, and youth. Pediatric clinical trial nursing requires a skill set tailored to the developmental needs of infants, children, and youth, including developmentally appropriate communication, child‑sensitive, comfort‑focused techniques that reduce fear and anxiety, and comprehensive family‑centred care. Pediatric clinical trial nursing expertise includes:
- Participant monitoring and assessment
- Investigational product administration (IV, oral, injection)
- Vital signs, anthropometrics, and clinical measurements
- Research sample collection
- Family education and support
- Protocol adherence to ensure safe, consistent implementation
Dedicated Research Phlebotomy
Children and youth participating in research benefit from:
- Skilled pediatric‑trained phlebotomists experienced in supporting young participants
- Reduced wait times supported by a dedicated research phlebotomy space within the CIU
- A calm, child‑friendly approach that helps reduce fear and discomfort
- Clinical and study bloodwork completed together during the same visit, minimizing additional blood draws and extra appointments
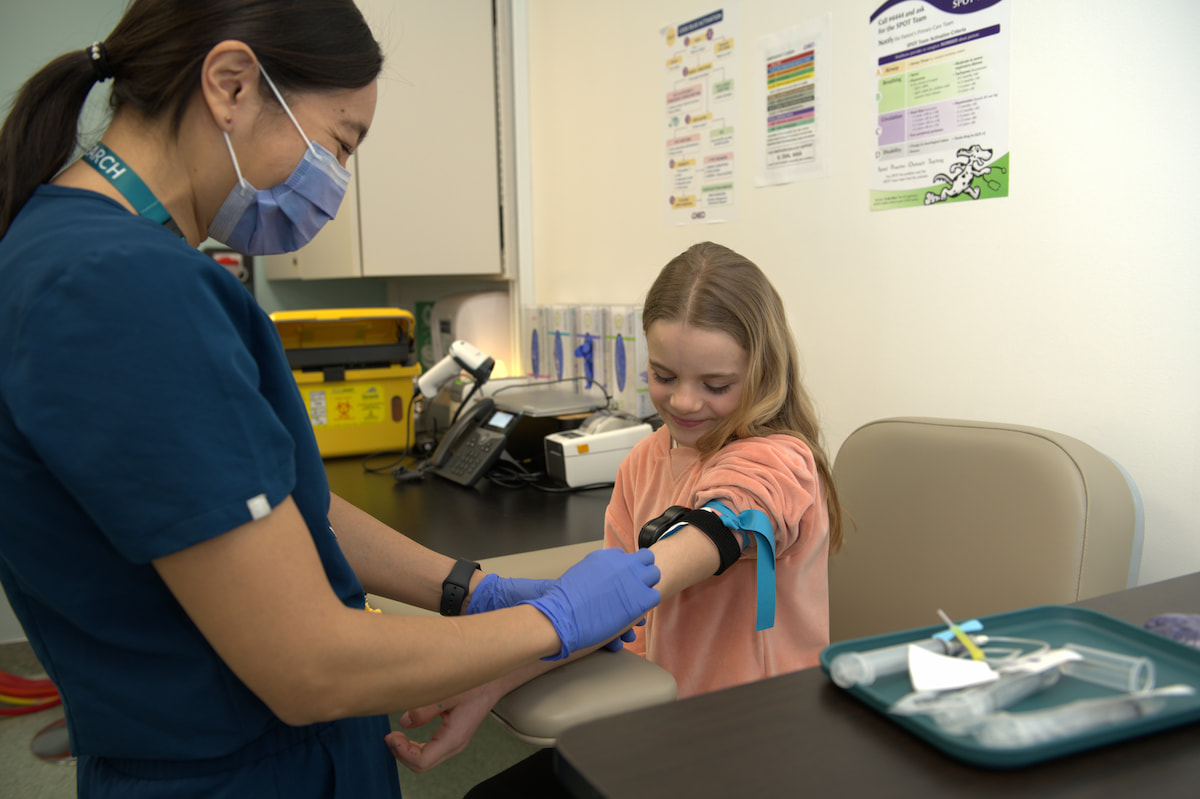
Clinical Research Space
The CIU offers:
- A private, single‑bed space designed to support uninterrupted pediatric trial visits
- Pediatric‑appropriate equipment tailored to the needs of infants, children, and youth
- A quiet, comfortable environment that helps reduce stress for children and families
- A controlled research setting that supports safe, consistent, and high‑quality study conduct
This dedicated clinical research space is used for both simple and complex trial procedures, ensuring flexibility while maintaining a child‑ and family‑centred experience.
Who We Support
Patients & Families
The safety, comfort, and experience of children and youth, as well as their families, are at the heart of the CIU. In the CIU, families can expect:
- Skilled pediatric clinical research staff
- Clear, developmentally appropriate communication and support
- A welcoming, family‑centred environment designed for children and youth
- Comfortable caregiver seating with space to work or rest during longer visits
Families interested in clinical trials are encouraged to speak with their CHEO care team.



Researchers
The CIU supports both investigator‑initiated and industry‑sponsored studies by providing:
- Centralized, dedicated clinical research space
- Infrastructure that strengthens feasibility, protocol adherence, and overall study quality
- Access to experienced pediatric clinical research nurses who provide practical guidance throughout trial planning and execution
- Mentorship from CIU staff and the Scientific Director to support investigators new to pediatric clinical trials
Industry Sponsors
The CHEO Research Institute is an experienced and highly engaged partner in pediatric clinical research, offering the specialized expertise, infrastructure, and operational support needed to conduct high‑quality, ethically sound trials involving children and youth. Industry partners benefit from:
- Dedicated pediatric clinical trial infrastructure, including purpose‑built space within the CIU
- Specialized pediatric clinical and research expertise, with staff skilled in age‑appropriate procedures, communication, and participant support
- Capacity to support a wide range of trial procedures and visit types, from simple assessments to complex, multi‑step protocol visits
- Streamlined coordination across clinical, research, and operational teams, reducing delays and enhancing site readiness
- A strong institutional commitment to research quality, participant safety, and regulatory compliance, ensuring trials are conducted to the highest standard
- Integrated services through the Clinical Trial Access Core, providing centralized expertise, harmonized processes, and consistent support throughout startup and execution
Sponsors interested in exploring collaboration opportunities are encouraged to contact our team to discuss capabilities, feasibility, and potential partnerships.
Contact Us
Reach out to learn more about the Clinical Trial Access Core, the Clinical Investigation Unit, or connect with our team at [email protected]

